|
11/21/2023 0 Comments Carbon periodic table weight 
Early work by Krouse established the wide natural range in the atomic weight of significant elements including carbon and sulfur. Roy Krouse created the Stable Isotope Laboratory in the Department of Physics and Astronomy in 1971. The University of Calgary has and continues to contribute substantially in the study of atomic weight variations. "We hope that chemists and educators will take this challenge as a unique opportunity to encourage the interest of young people in chemistry and generate enthusiasm for the creative future of chemistry." Fabienne Meyers, associate director of IUPAC. "Though this change offers significant benefits in the understanding of chemistry, one can imagine the challenge now to educators and students who will have to select a single value out of an interval when doing chemistry calculations," says Dr. For example, the standard atomic weights for fluorine, aluminum, sodium and gold are constant, and their values are known to better than six decimal places. "In other words, knowing the atomic weight can be used to decode the origins and the history of a particular element in nature," says Wieser who co-authored the report.Įlements with only one stable isotope do not exhibit variations in their atomic weights. However, its actual atomic weight can be anywhere between 32.059 and 32.076, depending on where the element is found. The changes to be made to the Table of Standard Atomic Weights have been published in Pure and Applied Chemistry and a companion article in Chemistry International.įor example, sulfur is commonly known to have a standard atomic weight of 32.065. The atomic weights of these 10 elements now will be expressed as intervals, having upper and lower bounds, reflected to more accurately convey this variation in atomic weight. In sports doping investigations, performance-enhancing testosterone can be identified in the human body because the atomic weight of carbon in natural human testosterone is higher than that in pharmaceutical testosterone. Isotopic measurements of nitrogen, chlorine and other elements are used for tracing pollutants in streams and groundwater. For example, precise measurements of the abundances of isotopes of carbon can be used to determine purity and source of food, such as vanilla and honey. Modern analytical techniques can measure the atomic weight of many elements precisely, and these small variations in an element's atomic weight are important in research and industry. This organization oversees the evaluation and dissemination of atomic-weight values. Michael Wieser, an associate professor at the University of Calgary, who serves as secretary of the International Union of Pure and Applied Chemistry's (IUPAC) Commission on Isotopic Abundances and Atomic Weights. As technology improved, we have discovered that the numbers on our chart are not as static as we have previously believed," says Dr. 
"For more than a century and a half, many were taught to use standard atomic weights - a single value - found on the inside cover of chemistry textbooks and on the periodic table of the elements. 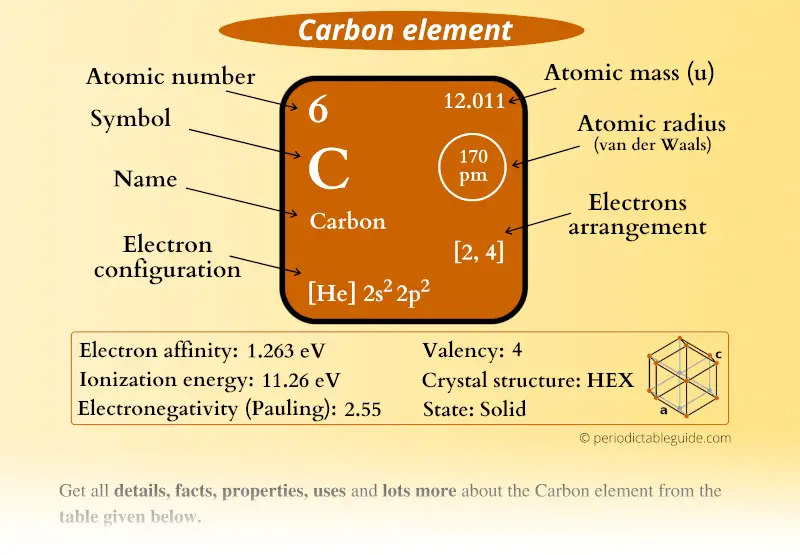 
The new table, outlined in a report released this month, will express atomic weights of 10 elements - hydrogen, lithium, boron, carbon, nitrogen, oxygen, silicon, sulfur, chlorine and thallium - in a new manner that will reflect more accurately how these elements are found in nature. view moreĬredit: Riley Brandt/University of Calgaryįor the first time in history, a change will be made to the atomic weights of some elements listed on the Periodic table of the chemical elements posted on walls of chemistry classrooms and on the inside covers of chemistry textbooks worldwide. He works with a thermal ionization mass spectrometer used to measure the isotope abundance of an element. Image: Michael Wieser, a professor at the University of Calgary, is contributing to changes to the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
